Photospectra is the first company in Canada and the USA to undertake a clinical trial of Photobiomodulation (PBM) for Dry AMD.
We expect PBM to be a major new treatment for dry AMD patients – safe, effective and affordable when proven in clinical studies. We are dedicated to proving the merits of PBM through ethical, responsible, well designed and well-executed clinical studies.
The TORPA trial was completed and presented at ARVO 2012. Further off label data has been presented internationally at ARVO 2016 in Seattle and Euretina 2016 in Copenhagen. This data has now been published in ACTA Ophthalmologica; a high impact, peer reviewed, international Ophthalmology journal.
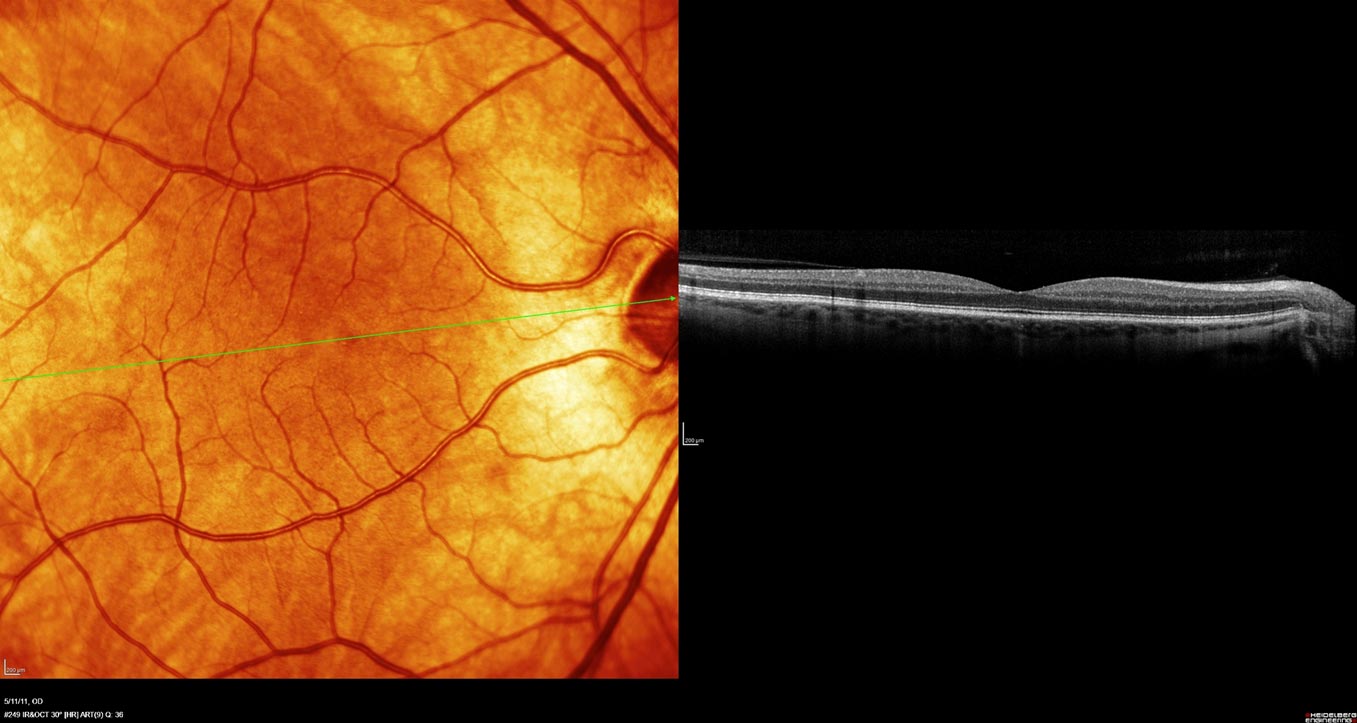
Photospectra has noted some very exciting anatomical changes of drusen reduction in association with vision improvement.
Company and Industry News
(More here)
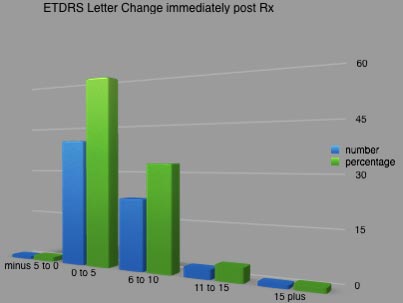
Interim results from the LIGHTSITE I clinical trial announced.
Interim results were presented at the American Academy of Ophthalmology (AAO) meeting in New Orleans USA, November 11-14 2017. LIGHTSITE I enrolled 30 dry AMD subjects who were randomized (1:1) to PBM treatment or sham treatment. “We have examined the interim data...
Resignation from LumiThera
Drs. Merry and Dotson resigned from the board of directors of Lumithera Inc. on April 3rd 2017. Both physicians no longer have an active management role within Lumithera Inc. They both however remain firm believers in the science and clinical application of...
He, along with Dr. Robert Dotson, have been pioneering the clinical application of Photobiomodulation in AMD and hope to provide a simple, non-invasive therapy for this enormous unmet need.
For more on Dr. Merry please read About Us.